Lecture NotesIsotopesIsotopes are same elements with different number of neutrons.
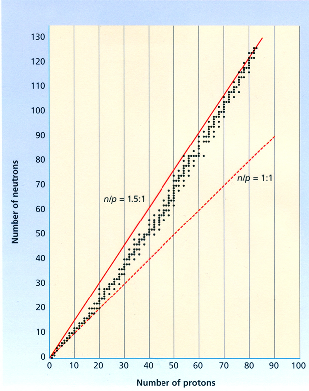
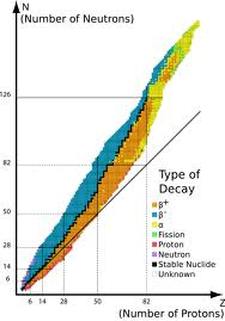
Radioactive isotopes are determined by the stability of the atom's nucleus (ratio of protons to neutrons). Many isotopes have a ratio of protons to neutrons that renders them unstable and, as a result, they are radioactive. Watch the Youtube video for detailed explanation of radioactive isotope. Band of StabilityResources: |
Nuclear Chemistry WorksheetsRead this web page,click on the lectures resources to read lecture notes, and watch the Youtube video, then complete the worksheets below. This will be classwork + homework. Parts of it will be done in class and part at home (you are expected to finish the part not finished in class).
Who is this lady?Find out more about this lady by clicking on the picture. For extra credit, find out how she contributed to nuclear chemistry.
| ||||||||||||||||||||||||
Nuclear Energy
Fusion vs FissionWhy should we care about nuclear chemistry? Many reasons but one of the foremost is the sun. Daily the sun, our star, provides energy needed to sustain this planet. Without sun, this planet called earth will perish. Well, that sun cannot exist without nuclear (fusion) reaction.
Nuclear Fusion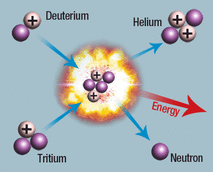
In the sun, hydrogen atoms combine to form helium atom through nuclear fusion. This process produces a lot of energy. Deuterium and Tritium are isotopes of hydrogen.
Nuclear Fission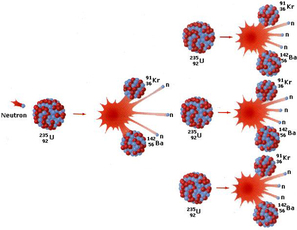
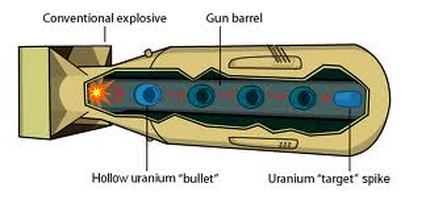
In both producing clean nuclear energy (compare to fossil fuel) and making nuclear weapons (atomic bombs),nuclear fission reaction is used. In nuclear fission reaction, a heavy nucleus splits spontaneously or on impact with another particle, with the release of large amounts of energy (which can create a chain reaction as seen picture above.
Video Link: Discovery Channel Nuclear Bomb
Type of Nuclear Decay Lecture & WorksheetDownload, read the lecture notes, then complete the worksheet (It's Greek to me).
|
Nuclear Energy Debate
| ||||||||||||||||||||||||||||||
| graphic_organizer.pdf | |
| File Size: | 353 kb |
| File Type: | |
| nuclear_energy_debate_.pdf | |
| File Size: | 238 kb |
| File Type: | |
| preassessment__rubric.pdf | |
| File Size: | 641 kb |
| File Type: | |
Resources
Lots More Information
Other Great Links
Video Links
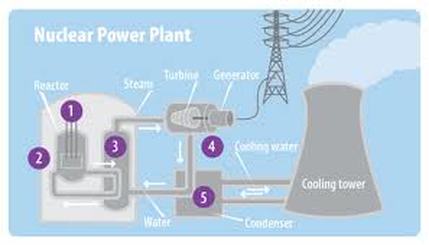
How does Nuclear Power Plants work?
Accident @ Fukushima Nuclear Power Plant
Radioactivity & ~YOU~
Writing Activity: Please read the "Modern Use of Radioactive Isotopes" and "Everyday Radiation Exposure" then, summarize each uses and summarize how you are exposed to radiation everyday.
Modern Use of Radioactive Isotopes*1. Smoke DetectorsChances are that a great number of homes have had one or more of these devices installed as an early warning system in case of fire. What most consumers don't know is that many of these units contain a small amount of americium-241 which emits alpha particles. By utilizing the radioactive properties of this material, smoke from a fire can be detected at a very early stage. Research about chemistry involved in smoke detectors.
2. Archaeological DatingAll biologic organisms contain a given concentration of carbon-14 isotope, we can use this information to help solve questions about when the organism died.
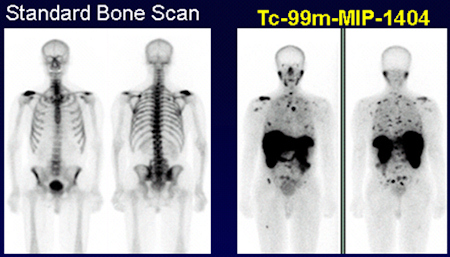
When an organism dies it has a specific ratio by mass of carbon-14 to carbon-12 incorporated in the cells of it's body. (The same ratio as in the atmosphere.) At the moment of death, no new carbon-14 containing molecules are metabolized, therefore the ratio is at a maximum. After death, the carbon-14 to carbon-12 ratio begins to decrease because carbon-14 is decaying away at a constant and predictable rate. Remembering that the half-life of carbon-14 is 5700 years, then after 5700 years half as much carbon-14 remains within the organism. 3. Medical Use (Nuclear Medicine)Bone imaging is an extremely important use of radioactive properties. Supposed a runner is experiencing severe pain in both shins. The doctor decides to check to see if either tibia has a stress fracture. The runner is given an injection containing 99Tcm (Technetium-99m). This radioisotope is a gamma ray producer with a half-life of 6 hours.
After a several hour wait, the patient undergoes bone imaging. At this point, any area of the body that is undergoing unusually high bone growth will show up as a stronger image on the screen. Therefore if the runner has a stress fracture, it will show up on the bone imaging scan. 4. Food IrradiationFood irradiation is a method of treating food in order to make it safer to eat and have a longer shelf life. This process is not very different from other treatments such as pesticide application, canning, freezing and drying. The end result is that the growth of disease-causing microorganisms.
Food irradiated by exposing it to the gamma rays of a radioisotope -- one that is widely used is cobalt-60. The energy from the gamma ray passing through the food is enough to destroy many disease-causing bacteria as well as those that cause food to spoil, but is not strong enough to change the quality, flavor or texture of the food. It is important to keep in mind that the food never comes in contact with the radioisotope and is never at risk of becoming radioactive
5. Agricultural Application - radioactive tracersRadioisotopes can be used to help understand chemical and biological processes in plants. This is true for two reasons: 1)radioisotopes are chemically identical with other isotopes of the same element and will be substituted in chemical reactions and 2)radioactive forms of the element can be easily detected with a Geiger counter or other such device.
Example:
A solution of phosphate, containing radioactive phosphorus-32, is injected into the root system of a plant. Since phosphorus-32 behaves indentically to that of phosphorus-31, the more common and non-radioactive form of the element, it is used by the plant in the same way. A Geiger counter is then used to detect the movement of the radioactive phosphorus-32 throughout the plant. This information helps scientists understand the detailed mechanism of how plants utilized phosphorus to grow and reproduce. |
Everyday Radiation Exposure*Regardless of what you do on a typical day, go to school, work, or stay home, in each case you are being exposed to radiation. Whether you know it or not you are being exposed to radioactivity everyday of your life.
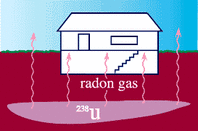
Nuclear radiation is all around us in the environment. Low-level radiation is found in the oceans and waterways, the rocks and soils, the plant materials and in the atmosphere surrounding the planet. The radiation that we are exposed to can be said to come from two sources, that which occur naturally and that which is due to the activities of man. Natural Radiation Terrestrial Radioactivity in nature comes from two main sources, terrestrial and cosmic. Terrestrial radioisotopes are found on the earth that came into existence with the creation of the planet. Although some are long gone, some radioisotopes take a long time to decay and become non-radioactive (on the order of hundreds of millions of years) and are still around today. By far, the largest contributor to our daily exposure of radiation is the natural world, and the major form of natural radiation is radon gas. Radon-222 is a naturally occuring decay product of uranium-238 which is commonly found in soils and rocks. Radon-222 is a gas which is odorless, colorless, tasteless and chemically nonreactive. As it escapes from the soils and rocks of which it is trapped, it enters the water we drink and the air we breath. Cosmic
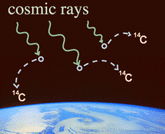
Another source of natural radiation comes from the interaction of cosmic rays with the earth's upper atmosphere. Cosmic rays permeate all of space and are composed of highly energized, positively charged particles as well as high energy photons. Approaching the earth at near the speed of light, most cosmic rays are blocked by the earth's protective atmosphere and magnetic field. As a byproduct of the interaction between cosmic rays (i.e. particles) and the atmosphere, many radioactive isotopes are formed such as carbon-14. Cosmic rays are also composed of high energy photons, and not all are prevented from reaching the earth's surface. It makes sense that the higher you are in altitude, the more you are exposed to cosmic radiation. Nuclear Radiation from Human Activities
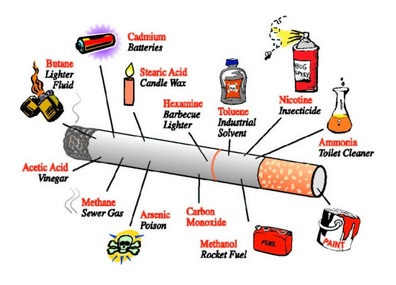
Although radioisotopes occur naturally in the environment, activities of humans have brought this radiation closer to us all.The human production of tobacco products introduces another way for us to get exposure to radiation. Smokers receive a dose of radiation from polonium-210 which is naturally present in tobacco. Smokers also receive an additional dose of radiation from the decay product of radon gas, polonium-218. Polonium-218 clings to aerosols such as tobacco smoke, and eventually winds up in the lungs. Once in the lungs, polonium decays by alpha particle emission and in the process may damage cells. Although this does not contribute radiation on a daily basis, the medical field has several ways of causing exposure to an individual. This exposure results from the attempt to diagnose fractures or cavities using x-rays, or to diagnose or treat cancer using injected radioisotopes. Patients are exposed to nuclear radiation in the diagnosis and treatment of cancer. Additionally, radiologists routinely use radioisotopes of technetium or thorium to diagnose heart disease. Radiation and Your Health*Any amount of radiation can be dangerous because of the potential effect that it has on living cells. Radiation can disrupt normal chemical processes of the cells, causing them to grow abnormally or to die. Cells that are altered by the radiation may go on to produce more abnormal cells - a process that could eventually lead to cancer.
At low doses, such as have been described here, cells are able to repair any damage rapidly. Any cells that die due to exposure can be replaced by the body. If one receives a very high dose, unlike any exposure mentioned here, the cells may not be able to be replaced fast enough and tissues or organs may fail to function properly. Guidelines for levels of exposure have been set by the Deparment of Energy as means of protecting the public. *Information from Duke University website. |