So Far....
We learned that chemistry is study of matter. Matter is made up of elements and element is made up of individual atoms. These atoms are made up of nucleus (protons & neutrons) and electrons. We looked at what happens when there is a change in the nucleus in the nuclear reaction. Now it's time to look at the electrons. How they behave will lead to different chemical bond formation. It's all about electrons here.
Chemical BondingPlease download the document below which contains lecture notes and some questions to check for your understanding.
|
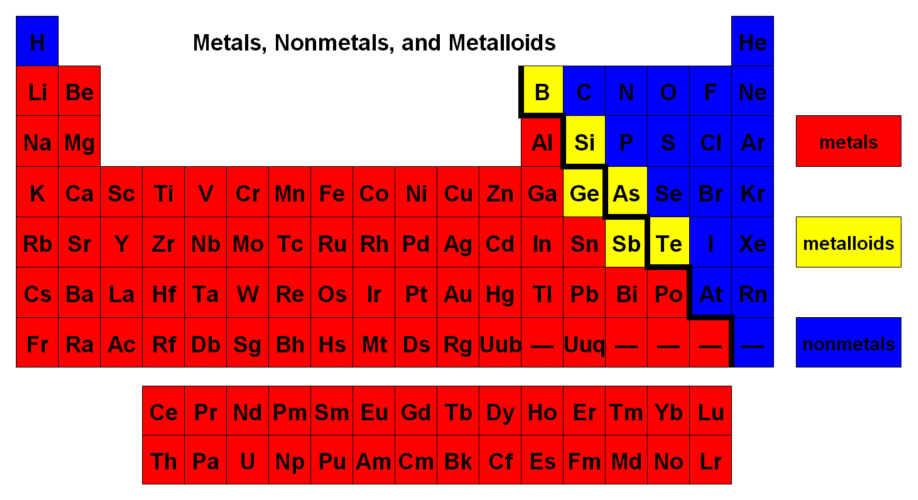
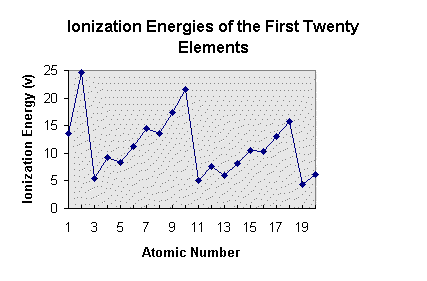
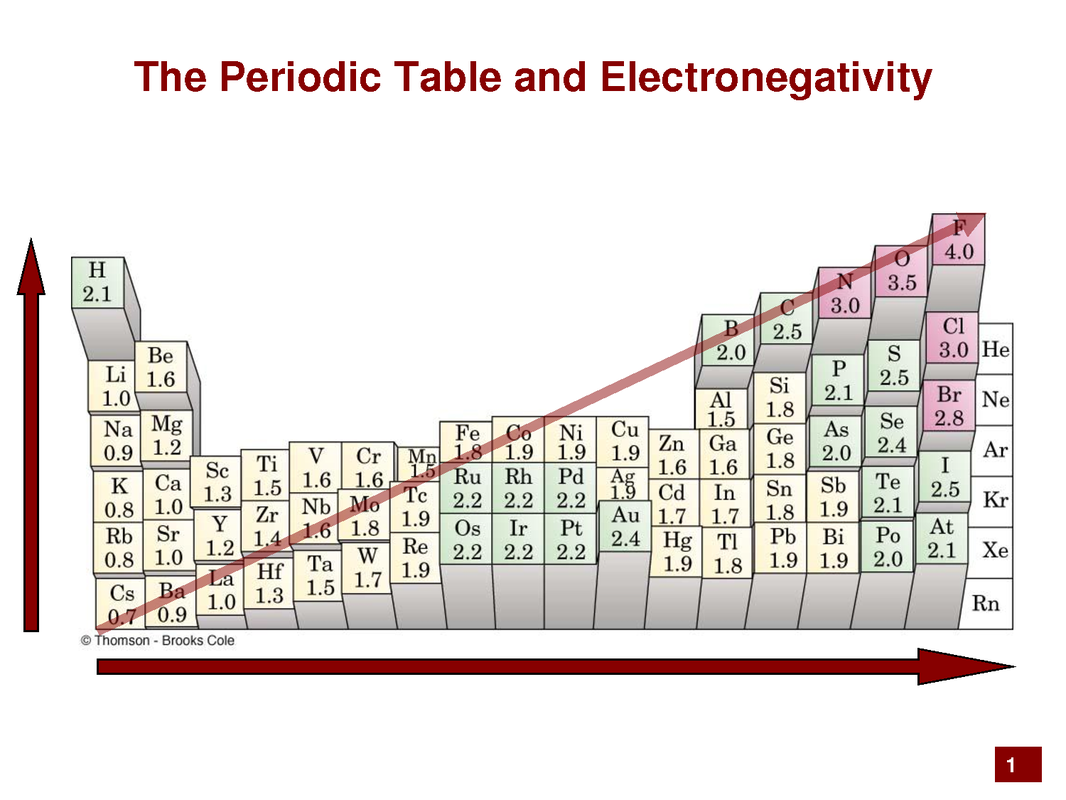
Periodic Table Trends.Download documents and complete. You will need graph paper to graph the data points first. Then answer the questions.
| ||||||||||||||||||||||||||||||||
Intramolecular forces
|
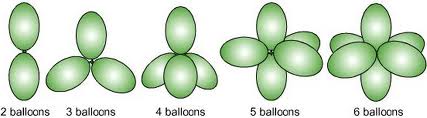
Within a molecule there is a bit of power struggle for electrons. The winner is the atom with greater electronegativity. Fluorine and its neighbors rule here creating polarity within a molecule (+ & - poles). This polarity gives rise to many unique properties.
Please read pg 88 - 91 in Bear Essentials of Polarity handout then answer questions on page 86.
|
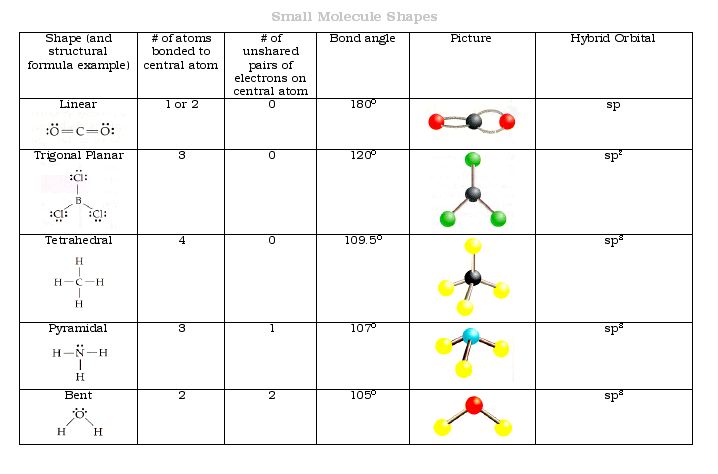
After completing the Bear Essential Worksheet, download the document below to complete 3D ball-and-stick model lab.
| ||||||||||||
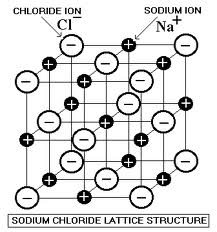
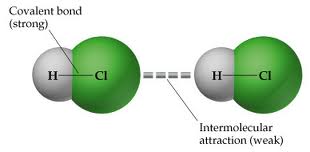
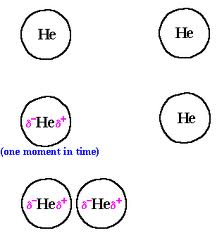
Intermolecular forces
There is a force between molecules. This force determined by polarity of each molecule. Complete the worksheet below to learn about the forces/attractions present between molecules.
| intermolecular_forces_worksheet.pdf | |
| File Size: | 159 kb |
| File Type: | |
Properties of water
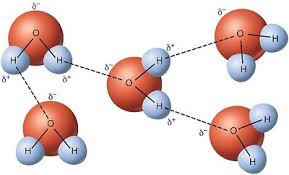
Hydrogen BondingDue to its polarity, water exhibits hydrogen bonding, the attraction between its negative O pole and its positive H pole. This hydrogen bonding allows for many unique properties in water. List of properties are on the right. At the end of they water unit, you will have to complete a demonstration quiz.
Due to this very property, water is less dense as solid. Most solid contracts (spatially) as it moves from liquid to solid phase but water keeps the space (does not contract) due to hydrogen bonding then solidifies. Hence, ice floats in water.
Water Property Videos |
1. High Heat of Vaporization
| ||||||
| tie_dye_lab.pdf | |
| File Size: | 238 kb |
| File Type: | |