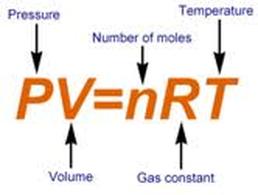
Ideal Gas LawThe formula above is the most important formula to remember for this particular unit. It shows the relationship between pressure, volume, temperature, and number of particles in dealing with gas molecules. Remember to use correct units.
Related Video & Article1. Rock me Avagadro - PBS
2. Gas Properties - PBS 3. Discovering Air 4. Mythbusters: Air Cylinder Rocket Laboratory Assignment1. Vacuum Bell Jar Experiment
2. Popcorn Lab 3. Gas Law Laboratory Practical 4. Jeopardy Game Review before the Test Gas Laws Mystery - Kilroy Caper
CR - FlatusRead the article and answer questions.
CR - SCUBA Diving
|
Lecture notes & Worksheet
Charles' Law describes the relationship between temperature and volume (at constant pressure).
Why does the can crush when place in cold water? What is happening to the gas inside the can? Use Charles' Law to explain this phenomenon. 
Boyle's Law describes the relationship between pressure and volume (at constant temperature).
Can you explain why the boiled egg goes into the glass bottle with a tiny mouth? Use Boyle's Law to explain this amazing event.
Mole Calculation
This is a fun subject in chemistry that might confuse some. If you are unsure how to calculate molar mass, please download the worksheet below and practice some more. It includes answers so check your work when you are done. Celebrate national mole day - 6:02 am 10/23.
Extra Credit - Everyday Examples of Gas Law
You observe gas law everywhere. Why? We are surrounded by gas (air). For example, car tires expand on a hot day. This can be explain by Charles' Law. As temperature rises, the volume of tire increases. For extra credit, find some examples of gas law around you (that's not already shared) then share with your classmates and your teacher. I look forward to your examples. | ||||||||||||||||||||||||||||||||||||||||||
Quick Review
|
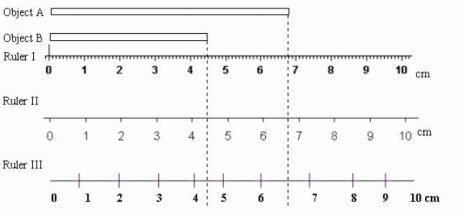
Significant Figures: any digit of a number that is known with certainty
Remember Sig. Fig. Rules 1. Rules for counting number of significant figures 2. Rules for addition & subtraction 3. Rules for multiplication and division
|
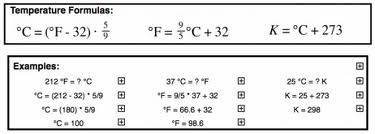
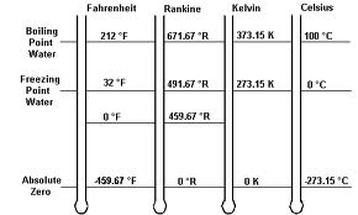
Temperature unit Conversion: Be able to convert between Farenheit, Celsius, and Kelvin.
Scientific notation: a short way to write very large or very small numbers. It is written as the product of a number between 1 and 10 and a power of 10.
| ||||||||||||||||||