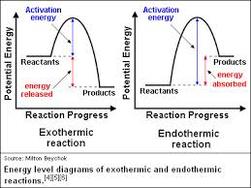
Exothermic vs. Endothermic ReactionIn speaking of thermodynamics, chemical reactions can be divided into two categories: Exothermic, heat-producing or Endothermic, heat-absorbing reactions.
Good examples of these are hot packs and cold packs. One type of instant hot packs use the exothermic reaction of rust formation and another uses the crystallization of super-saturated solution of sodium acetate. In the former, when the outer plastic bag is opened the iron powder (and catalysts) reacts with oxygen forming rust. More you shake it hotter it gets as more oxygen molecules react with iron powder to form iron oxide, rust. Instant cold packs contain two things: a white powder and a weaker inner bag of water. When the pack is punched, the inner bag breaks so water mixes with the white powder, a substance called ammonium nitrate. The reaction is an endothermic absorbing heat or as non-chemists say, "it gets cold." |
Measuring Calories in FoodCalorie is of heat energy. In the laboratory exercise in class we will be measuring the calorie produced by cheetos.
Calories in Cheetos (Label)
CR - Cold Packs & Hot Packs
| ||||||||||||||||||||
|
|