LD50
An LD50 is a standard measurement of acute toxicity that is stated in milligrams (mg) of substance per kilogram (kg) of body weight. An LD50 represents the individual dose required to kill 50 percent of a population of test animals (e.g., rats, fish, mice, cockroaches). Because LD50 values are standard measurements, it is possible to compare relative toxicities among substance. The lower the LD50 dose, the more toxic the substance. It take very little to kill hence, very toxic.
Example: A pesticide with an LD50 value of 10 mg/kg is 10 times more toxic than a pesticide with an LD50 of 100 mg/kg.
After studying the chart, use it to complete the worksheet. Chart source: https://ceufast.com/blog/the-lethal-doses-of-55-substances
Example: A pesticide with an LD50 value of 10 mg/kg is 10 times more toxic than a pesticide with an LD50 of 100 mg/kg.
After studying the chart, use it to complete the worksheet. Chart source: https://ceufast.com/blog/the-lethal-doses-of-55-substances
|
| ||||||||||||
How to Prepare Chemical Solution
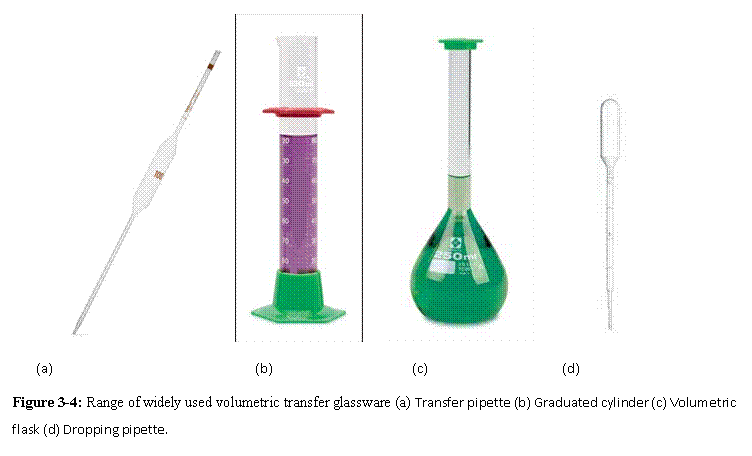
Glassware used in solution preparation |
Basic termsSolute - The substance which dissolves in a solution
Solvent - The substance which dissolves another to form a solution. For example, in a sugar and water solution, water is the solvent; sugar is the solute. Solution - A mixture of two or more pure substances. In a solution one pure substance is dissolved in another pure substance homogenously. For example, in a sugar and water solution, the solution has the same concentration throughout, ie. it is homogenous. Mole - A fundamental unit of mass (like a "dozen" to a baker) used by chemists. This term refers to a large number of elementary particles (atoms, molecules, ions, electrons, etc) of any substance. 1 mole is 6.02 x 1023 molecules of that substance. (Avogadro's number).M
| ||||||